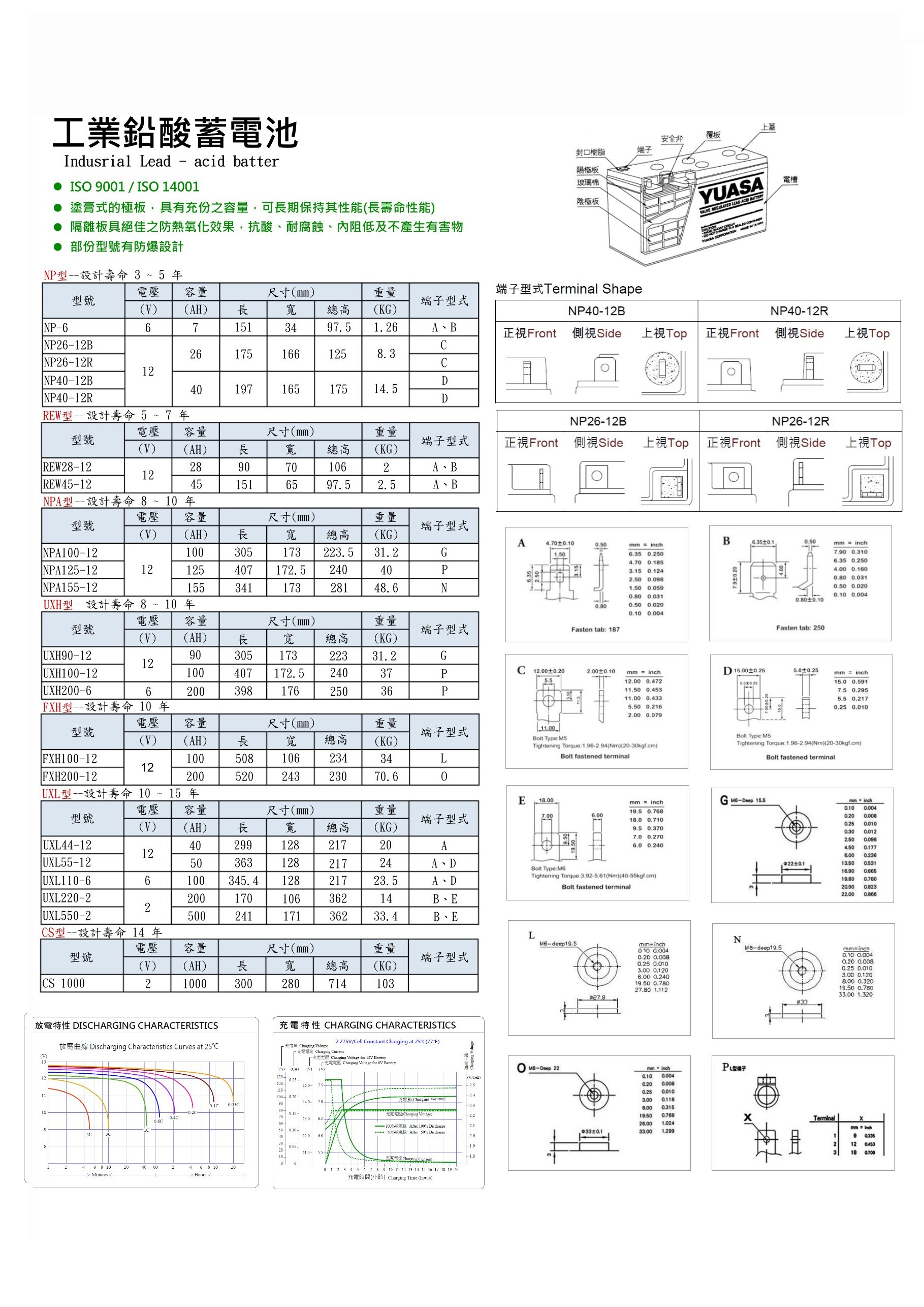
LEAD ACID BATTERY
2.There's two types of the lead-acid battery, flooded and vented lead acid battery.
3.The former needs regular acid-fluid injection to maintain,and the latter is the non-maintenance type but still needs regularly battery detection.
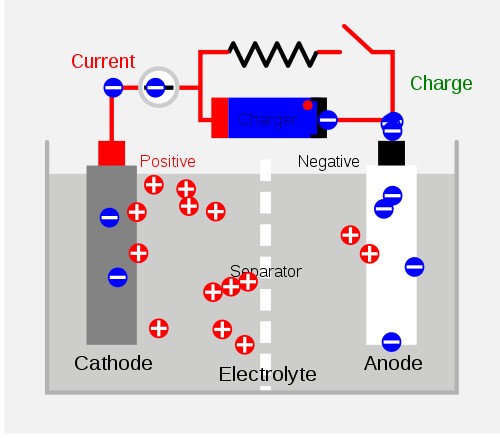
Lead-acie batteries
Lead-acid batteries are one of the most common secondary batteries, used primarily for storing large cell potential. These are commonly found in automobile engines. Its advantages include low cost, high voltage and large storage of cell potential; and disadvantages include heavy mass, incompetence under low-temperatures, and inability to maintain its potential for long periods of time through disuse. The reactions of a lead-acid battery are shown below:
Discharging:Pb(s)+SO4 2- (aq) → PbSO4(s)+2e-
Charging:PbSO4(s)+2e- → Pb(s)+SO4 2-
anode :
Discharging:PbO2(s)+4H+(aq)+SO4 2-(aq)+2e → PbSO4(s)+2H2O(l)
Charging:PbSO4(s)+2H2O(l) → PbO2(s)+4H+(aq)+SO4 2-(aq)+2e
overall :
Discharging:PbO2+Pb+2H2SO4 → 2PbSO4+2H2O
Charging:2PbSO4+2H2O → PbO2+Pb+2H2SO4
● safely sealed
During the normal operation, electrolyte will not leak from the terminal or the shell of the battery.The fluid-absorbing separator can isolate the acid fluid. With no free acid fluid inside therefore the battery can safely place to any place.
● air outlet system
When the internal pressure surpasses the normal standard, the VRLA battery will release extra gas and automativly sealed again to make sure there's no gas inside the battery.
●維護簡單 Easy to maintain
由於氣體複合系統使產生的氣體轉化成水,在使用VRLA電池的過程中不需要加水。
Because of the gas that made by gas compound system trun in to water, therefore in the process using the VRLA battery doesn't take water.
● Long using life
With the corrosion resitance lead-calcium alloy board the using time can up to 10 to 15 years.
● High quality,high relability
With the advanced produce tecknique and the strict quality control system can keep the quality of the VRLA battery steady . The voltage and the capacity run the 100% test on-line.